News
Mar 4, 2024
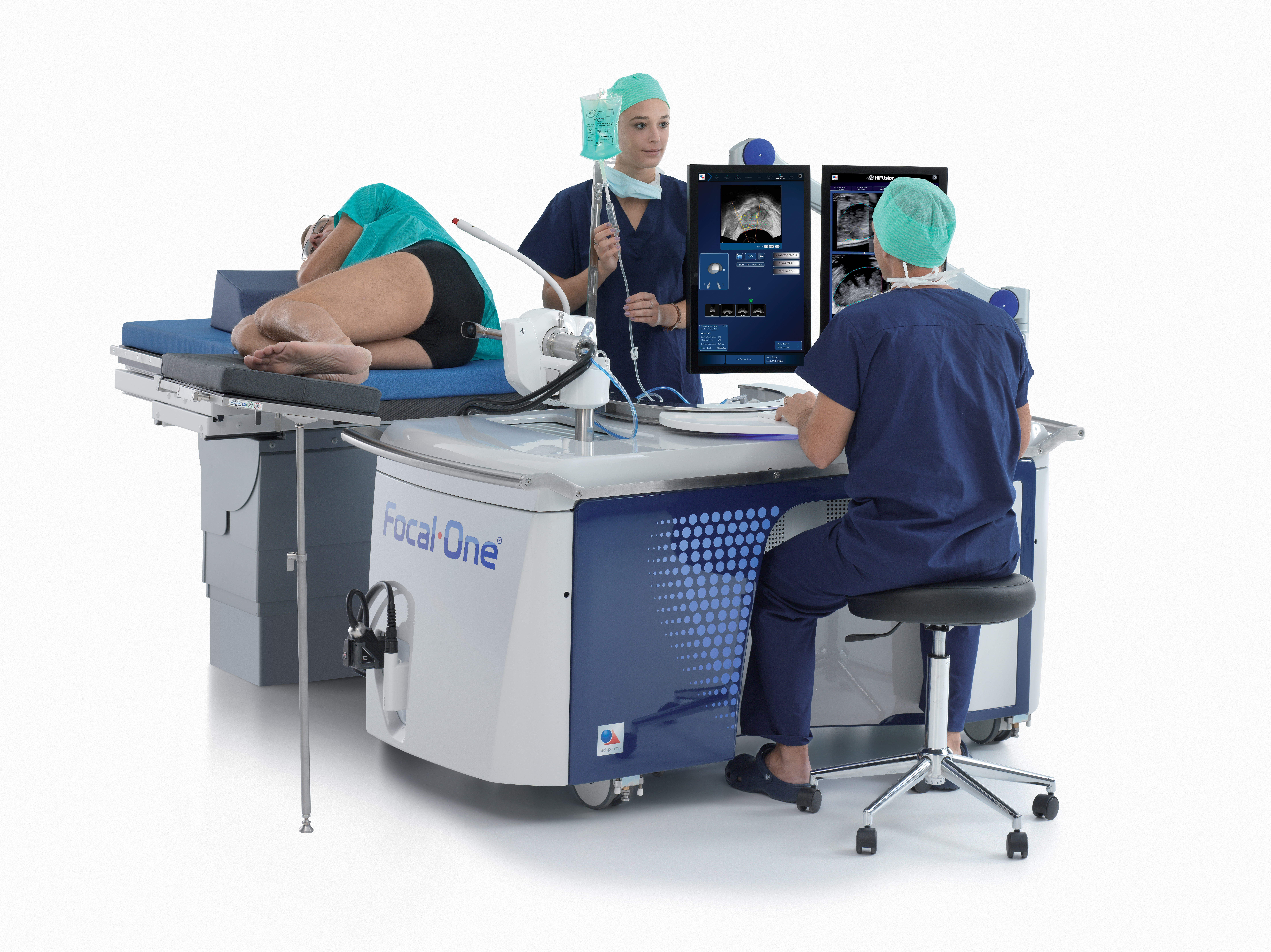
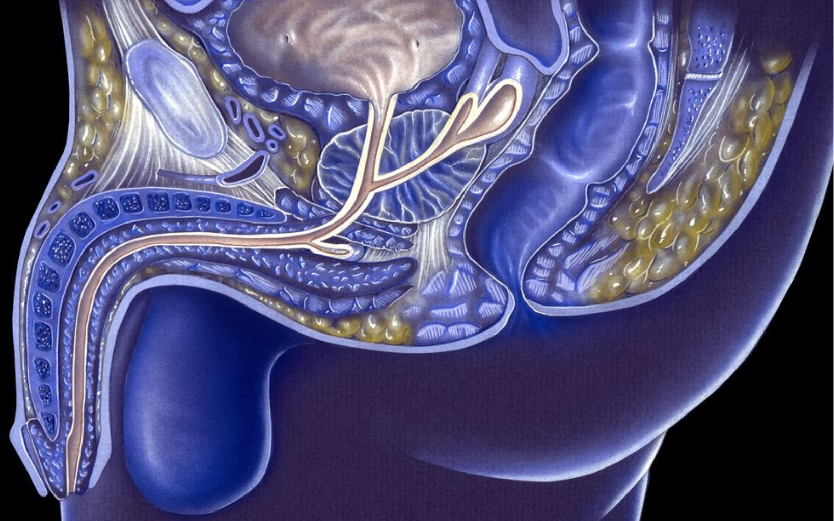
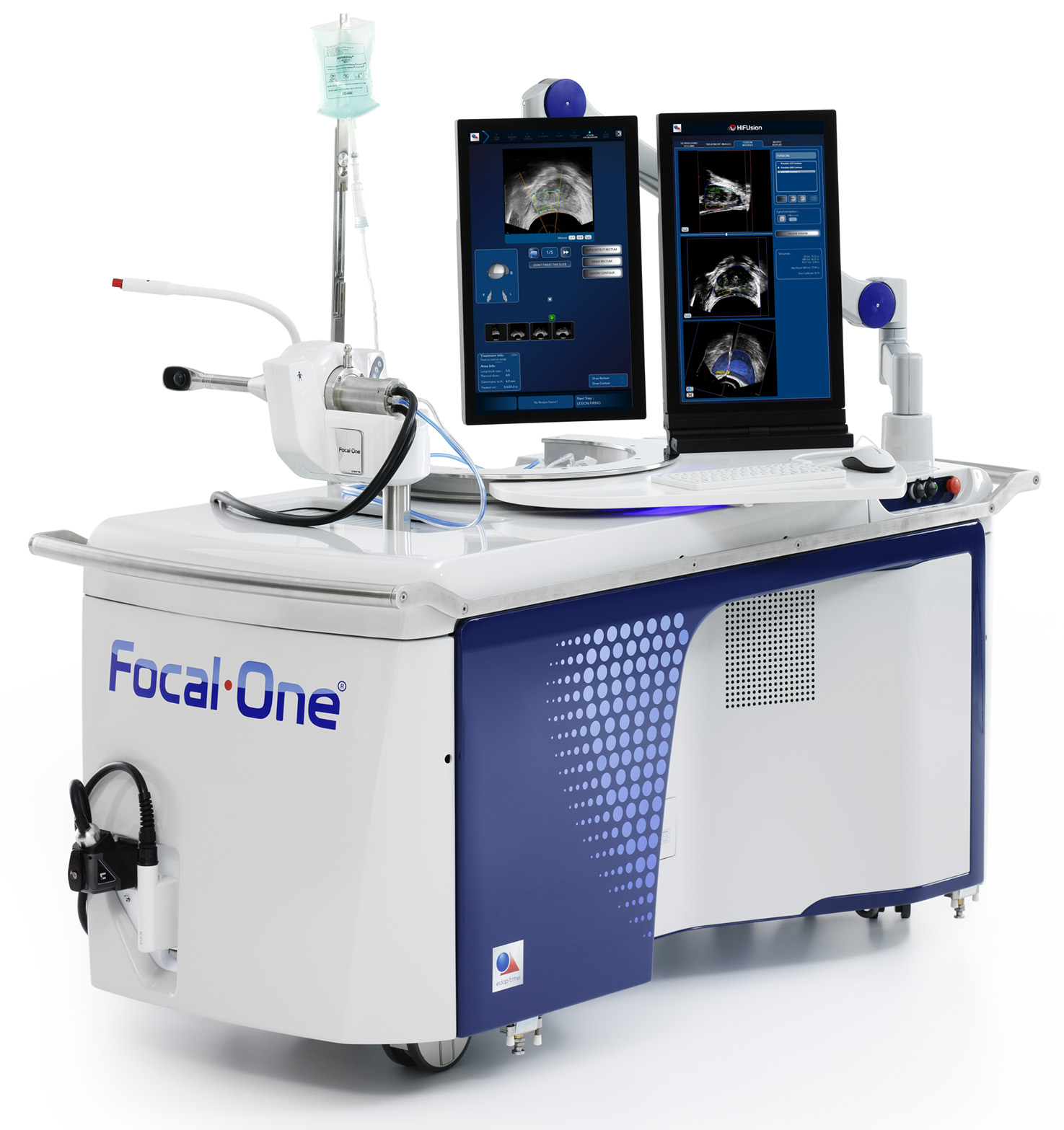
EDAP Announces FDA Breakthrough Device Designation for Focal One in the Treatment of Deep Infiltrating Rectal Endometriosis
EDAP Announces FDA Breakthrough Device Designation for Focal One in the Treatment of Deep Infiltrating Rectal Endometriosis
Read more
Jul 26, 2023
EDAP Announces Reimbursement Approval in Switzerland for the Use of High-Intensity Focused Ultrasound (HIFU) in the Treatment of Prostate Cancer
EDAP TMS SA (Nasdaq: EDAP) (“the Company”), the global leader in robotic energy-based therapies, today announced that it has received reimbursement approval in Switzerland for the use of High-Intensity Focused Ultrasound (HIFU) in the treatment of prostate cancer. This reimbursement took effect on July 1, 2023.
Read more
Apr 21, 2023
EDAP Announces Major Presence at Upcoming Annual Meeting of the American Urological Association (AUA)
Meeting to Showcase EDAP’s Focal One® Robotic High Intensity Focused Ultrasound (HIFU) Platform for the Management of Prostate Cancer
Read more
Apr 11, 2023
EDAP Announces Live Procedure Broadcast of Focal One High Intensity Focused Ultrasound (HIFU) to Global Clinical Audience
EDAP TMS SA, a global leader in robotic energy-based therapies, today announced that the Company will host a webcast that will feature a live procedure broadcast of its Focal One HIFU as a potential treatment for ablating cancer tissue in patients who are diagnosed with prostate cancer. The webcast is scheduled for Tuesday, April 18th at 12:00 p.m. PDT (3:00 p.m. EDT).
Read more
Mar 30, 2023
EDAP Announces Leadership Succession Plan to Develop Global Group Strategy
EDAP Announces Leadership Succession Plan to Develop Global Group Strategy
Read more
Mar 30, 2023
EDAP Reports Record Fourth Quarter and Full-Year 2022 Results
EDAP TMS SA, a global leader in robotic energy-based therapies, announced today unaudited financial results for the fourth quarter and full-year 2022
Read more
Feb 14, 2023
EDAP Announces Record Preliminary Fourth Quarter and Full-Year 2022 Revenue
EDAP TMS SA, a global leader in robotic energy-based therapies, today announced preliminary unaudited fourth quarter and full year 2022 revenue and other financial highlights.
Read more
Jan 31, 2023
EDAP Announces Positive Results from Phase 2 Study Evaluating Therapeutic for the Treatment of Rectal Endometriosis
EDAP TMS SA, the global leader in robotic energy-based therapies, today announced positive clinical results from the Endo-HIFU-R1 Phase 2 study evaluating the safety of therapeutic high-Intensity focused ultrasound (HIFU) for the treatment of rectal endometriosis. Results from the study were presented by study coordinator, Pr. Gil Dubernard of Croix Rousse University Hospital (Lyon, France), on Friday, January 27, at the Paris Santé Femmes French Congress1 (Lille, France).
Read more
Jul 18, 2022
EDAP Announces Focal One® HIFU Reimbursement Raised
EDAP Announces Focal One® HIFU Reimbursement Raised to Urology APC Level 6 Under CMS Outpatient Prospective Payment System (OPPS) Proposed Rule for CY23
Read more
Jun 24, 2022
EDAP’s Focal One® HIFU to be Showcased at Two Major Robotics & Urology International Congresses
EDAP’s Focal One® HIFU to be Showcased at Two Major Robotics & Urology International Congresses
Read more
Jun 14, 2022
EDAP holds a Symposium at 6th European Endometriosis Congress
Prof. Gil Dubernard, Head of the Gynecology Department at Croix-Rousse University Hospital, Lyon, France, will deliver a presentation at the 6th European Endometriosis Congress
Read more
May 5, 2022
EDAP to Showcase its Focal One® Platform at the Annual Meeting of the American Urological Association (AUA)
EDAP to Showcase its Focal One® Platform at the Annual Meeting of the American Urological Association (AUA)
Read more
Apr 12, 2022
EDAP Provides Corporate Update on US Focal One® Installation Growth
Company announces Focal One® placements at three renowned academic institutions in March
Read more
Oct 7, 2021
Beyond Death Valley: Medtech Veteran Breathes New Life into HIFU Company
Ryan Rhodes, new CEO of EDAP USA was featured in MDDI this week. Rhodes hales from Intuitive Surgical where he helped build the DaVinci Robot brand. He compares HIFU to the early days of robotic surgery: “The HIFU market is still in its infancy, and Rhodes said it reminds him of his early days at Intuitive Surgical, when the Irvine, CA-based robotics company was focused on prostate cancer, specifically radical prostatectomy, which was also a very young market at the time.”
Read more
Sep 7, 2021
EDAP's HIFU Progress Is Obscured By The Pandemic, But It's Real
Very positive article on EDAP’s future and its advances on #HIFU, written by Seeking Alpha.
Read more
Jun 15, 2021
EDAP Announces Hiring of MedTech Industry Veteran Ryan Rhodes as CEO of Company’s U.S. Subsidiary
LYON, France, June 15, 2021 -- EDAP TMS SA, the global leader in robotic energy-based therapies, announced today that the company has hired medical technology industry veteran Ryan Rhodes as Chief Executive Officer of EDAP Technomed, the Company’s U.S. subsidiary.
Read more
May 9, 2021
In the C-Suite with Marc Oczachowski, CEO, EDAP TMS
We venture to Lyon, France to learn how Marc and his team fresh off a $28 million raise are setting the stage to dominate High Intensity Focused Ultrasound (HIFU) for prostate cancer.
Read more
Mar 3, 2021
EDAP Selects Leading Reimbursement Consultancies MTP and Argenta Advisors to Expand Market Access and Coverage of HIFU in the US
EDAP TMS, the global leader in robotic energy-based therapies, announces today that the company has selected Medical Technology Partners (MTP) and Argenta Advisors, two leading reimbursement consultancies, to further expand coverage, accelerate market access and secure future payment rates for the company’s Focal One high intensity focused ultrasound (HIFU) procedure in the US. The announcement follows the establishment of a Category 1 CPT code, and physician reimbursement for HIFU, that went into effect on January 1, 2021.
Read more
Feb 18, 2021
The Future Is Now: Marc Oczachowski of EDAP TMS On How Their Technological Innovation Will Shake Up Healthcare
Marc Oczachowski is Chairman of the Board and Chief Executive Officer of EDAP TMS, a high-tech medical company which develops, manufactures markets minimally invasive therapeutic ultrasound solutions for urology, tumor removal, localized prostate cancer, and infectious diseases.
Read more
Jan 14, 2021
Positive 24 month interim results of French Urology Association (AFU) sponsored clinical trial study comparing HIFU to radical prostate surgery
Positive 24 month interim results of French Urology Association (AFU) sponsored clinical trial study comparing HIFU to radical prostate surgery: results on 3,329 patients demonstrate significant difference in efficacy in favor of HIFU versus Radical Prostatectomy (RP).
Read more
Dec 17, 2020
By bringing a safe, ambulatory and non-invasive solution to patients with #prostate conditions, watch how EDAP TMS and #HIFU users are handling the situation with COVID-19 when elective procedures are currently being put on-hold.
EDAP TMS and #HIFU users are handling the situation with COVID-19 when elective procedures are currently being put on-hold.
Read more
Dec 9, 2020
Final US 2021 reimbursement rules for High Intensity Focused Ultrasound (HIFU)
U.S. Centers for Medicare and Medicaid Services (CMS) has issued its final rules establishing, for the first time, a Category 1 CPT code which facilitates reimbursement for the ablation of malignant prostate tissue with HIFU technology, effective January 1, 2021.
Read more
Sep 3, 2020
First patients treated in phase 2 study of Focal One for the treatment of deep invasive endometriosis
the first two patients have been treated in the company’s Phase 2 clinical trial assessing Focal One® High Intensity Focused Ultrasound (HIFU) as a potential treatment for deep rectal endometriosis. A total of 38 women will be enrolled in the study and treated at five major hospitals and assessed over a six-month follow-up period.
Read more
Aug 25, 2020
Approval received for phase II study evaluating HIFU for deep invasive endometriosis
EDAP TMS has received approval from French health authorities to initiate a Phase II multicenter clinical trial evaluating its Focal One® HIFU to treat deep rectal endometriosis.
Read more
Aug 18, 2020
Two publications highlighting favorable HIFU focal therapy outcomes in journal of urology
Studies conducted at the University of Southern California and University of Miami represent the first U.S. publications detailing successful partial-gland ablation using high intensity focused ultrasound therapy to treat prostate cancer patients since FDA cleared the technology in 2015.
Read more
Aug 18, 2020
Two publications highlighting favorable HIFU focal therapy outcomes in journal of urology
Studies conducted at the University of Southern California and University of Miami represent the first U.S. publications detailing successful partial-gland ablation using high intensity focused ultrasound therapy to treat prostate cancer patients since FDA cleared the technology in 2015.
Read more
Jul 15, 2020
EDAP Announces Recent Focal One Sales Successes
Company completes four Focal One sales, including three bundled sales that combine Focal One® with Exact Imaging’s ExactVu micro ultrasound imaging solution.
Read more
May 12, 2020
EDAP announces exclusive worldwide distribution rights to EXACT Imaging's Micro-Ultrasound Technologies
EDAP announces exclusive worldwide distribution rights to EXACT Imaging's Micro-Ultrasound Technologies
Read more
Apr 23, 2020
B Riley FBR Initiates Research Coverage on EDAP TMS
Last week Andrew D’Silva of B Riley FBR launched research coverage on EDAP TMS with a buy-rating and price target at $5.50.
Read more
Mar 17, 2020
EDAP TMS about COVID-19
Update on COVID-19 and the Potential Impact to Operations
Read more
Apr 1, 2019
Focal One's precision means qualifying prostate cancer patients can forego prostate removal surgery and avoid life-altering side effects including incontinence and impotence
The University of Chicago Medicine became the first medical center in the Midwest to offer focal therapy to men with localized prostate cancer, using Focal One, the only advanced robotic ultrasound technology available to target and ablate diseased prostate tissue.
Read more
Mar 4, 2019
Houston Methodist's Reinforces EDAP's Position as a Leading HIFU Innovator
Houston Methodist Hospital, has acquired Focal One®, the most advanced HIFU technology available today
Read more
Jan 23, 2019
Maimonides Medical Center acquires Focal One Technology
Maimonides Medical Center acquires Focal One Technology
Read more
Jan 14, 2019
Two additional Focal One devices to two leading hospitals in Brazil
Hospital 9 de Julho and Hospital Moriah join growing roster of highly-regarded hospitals in Brazil offering Focal One
Read more
Jan 3, 2019
John Wayne Cancer Institute First US Medical Center to Acquire EDAP's Breakthrough Focal One Technology for Prostate Tissue Ablation
John Wayne Cancer Institute (JWCI) at the Providence Saint John's Health Center in Santa Monica, offer focal therapy to patients with localized prostate cancer using Focal One
Read more
Dec 12, 2018
Oncology Live - Urologists in Cancer Care
Focal Therapy Evolves Into More Precise Treatment
Read more
Nov 21, 2018
As the Wife of a Prostate Cancer Survivor
As the Wife of a Prostate Cancer Survivor I’m Eternally Grateful for Colorado’s Early Blood Draw
Read more
Oct 26, 2018
A New Treatment for Localized Prostate Cancer Lets Men Keep the Sexual Passion in Their Relationships
A New Treatment for Localized Prostate Cancer Lets Men Keep the Sexual Passion in Their Relationships
Read more
Oct 24, 2018
Urologists, uro-oncologists and urology department chairs from renowned American universities will spend a day at Jewish General Hospital
Urologists, uro-oncologists and urology department chairs from renowned American universities will spend a day at Jewish General Hospital
Read more
Oct 3, 2018
HIFU Prostate Procedure Helps NASA Safety Engineer Protect His Health
HIFU Prostate Procedure Helps NASA Safety Engineer Protect His Health
Read more
Sep 27, 2018
EDAP TMS SA the global leader in therapeutic ultrasound, today announced the installation of the third Focal One device in Brazil at the renowned Albert Einstein Center of Oncology,
EDAP TMS SA the global leader in therapeutic ultrasound, today announced the installation of the third Focal One device in Brazil at the renowned Albert Einstein Center of Oncolog
Read more
Sep 26, 2018
Prostate Cancer Leads Aerospace Biomedical Technician to Discover High-Tech HIFU
Charles McFarland shares his experience with High Intensity Focused Ultrasound treatment of prostate cancer.
Read more
Sep 20, 2018
EDAP announces Installation of First Focal One HIFU device in South Korea
EDAP announces Installation of First Focal One HIFU device in South Korea
Read more
Jul 30, 2018
Alternative Treatment for Localized Prostate Cancer: Three Men’s Perspectives
Alternative Treatment for Localized Prostate Cancer: Three Men’s Perspectives
Read more
Jul 24, 2018
Less-invasive prostate cancer treatment receives FDA clearance
Less-invasive prostate cancer treatment receives FDA clearance
Read more
Jul 24, 2018
Boomer Times Presents: Anita Finley with Clifford Gluck, MD, urologist
Boomer Times Presents: Anita Finley with Clifford Gluck, MD, urologist
Read more
Jun 20, 2018
University of Miami at Forefront of Prostate Cancer Treatment
University of Miami at Forefront of Prostate Cancer Treatment
Read more
Jun 7, 2018
EDAP Announces FDA Clearance for Focal One
EDAP Announces FDA Clearance for Focal One
Read more
May 24, 2018
HIFU Procedure Keeps High School Ref in the Game after Prostate Cancer Diagnosis
HIFU Procedure Keeps High School Ref in the Game after Prostate Cancer Diagnosis
Read more
Apr 9, 2018
Focal Ultrasound Prostate Ablation Shows Promising Results with Fewer Side Effects
Focal Ultrasound Prostate Ablation Shows Promising Results with Fewer Side Effects
Read more
Apr 9, 2018
Database to follow patients undergoing partial-gland HIFU
Database to follow patients undergoing partial-gland HIFU
Read more
Apr 8, 2018
Point: Is HIFU for low-risk prostate Ca ready for prime time?
Point: Is HIFU for low-risk prostate Ca ready for prime time?
Read more
Apr 6, 2018
In a First, US Insurer Covers HIFU for Prostate Cancer
In a First, US Insurer Covers HIFU for Prostate Cancer
Read more
Apr 3, 2018
EDAP Reports First US Cohort Study Results of Focal HIFU Prostate Ablation Shows Promising Outcomes
EDAP Reports First US Cohort Study Results of Focal HIFU Prostate Ablation Shows Promising Outcomes
Read more
Mar 28, 2018
New prostate cancer treatment found more effective than previous methods
New prostate cancer treatment found more effective than previous methods
Blair's doctor Dipen Parekh from the University of Miami School of Medicine says that the treatment is new and effective
Read more
Mar 28, 2018
New Prostate Cancer Treatment Is A Real Life Saver
New Prostate Cancer Treatment Is A Real Life Saver
Read more
Mar 26, 2018
Major US insurer to cover use of HIFU for treatment of radiorecurrent prostate cancer
Major US insurer to cover use of HIFU for treatment of radiorecurrent prostate cancer
Read more
Mar 21, 2018
EDAP Announces CIGNA as First Major U.S. Private Health Insurer to Cover HIFU for Prostate Procedure
EDAP Announces CIGNA as First Major U.S. Private Health Insurer to Cover HIFU for Prostate Procedure
Read more
Mar 6, 2018
Top Universities Join Forces to Launch First U.S. Based HIFU Patient Registry for Prostate Treatment
Top Universities Join Forces to Launch First U.S. Based HIFU Patient Registry for Prostate Treatment
Read more
Mar 5, 2018
How to Build a Business Case for Medical Devices Awaiting Reimbursement
How to Build a Business Case for Medical Devices Awaiting Reimbursement
Read more
Mar 2, 2018
Focusing On You - Interview with Dr. Dipen Parekh
Focusing On You - Interview with Dr. Dipen Parekh
Read more
Jan 26, 2018
Men with Prostate Cancer May Not Know All of Their Treatment Options and Risks
Men with Prostate Cancer May Not Know All Of Their Treatment Options and Risks
Read more
Jan 23, 2018
They may have cancer. But they’ll be at the starting line.
They may have cancer. But they’ll be at the starting line.
Read more
Jan 11, 2018
New Medical Technology Gives Urologists a More Precise Method to Target Diagnosed Areas of the Prostate
New Medical Technology Gives Urologists a More Precise Method to Target Diagnosed Areas of the Prostate
We asked Dr. Cary Robertson, M.D. from Duke University Medical Center three questions on the latest development of a new medical technology that gives urologists a more precise method for treating prostate cancer.
Read more
Jan 7, 2018
Impact: Opio Speaks with Dr. Ivan Grunberger about Prostate Cancer
Impact: Opio Speaks with Dr. Ivan Grunberger about Prostate Cancer
Read more
Jan 2, 2018
30 secondes interview with Dr. Brian Miles on KTRH (iHeart Radio)
30 secondes interview with Dr. Brian Miles on KTRH (iHeart Radio)
Read more
Dec 21, 2017
How to build a business case for medical devices awaiting reimbursement
In 2015, Houston Methodist Hospital made the decision to invest in a medical technology that had not yet been approved by the US Food & Drug Administration.
Read more
Dec 18, 2017
Prostate cancer therapy gives patients a different approach
Rochester, N.Y. (WHAM) - A new therapy is giving prostate cancer patients in Rochester an alternative to traditional cancer treatment.
Read more
Dec 17, 2017
A Health Screening Saved My Life
My Journey with Prostate Cancer Treatment
Read more
Dec 12, 2017
Prostate Cancer 101: What All Senior Men Should Know
Prostate cancer is the third most common type of cancer. It affects over 3 million men in the United States – most of them seniors.
Read more
Nov 29, 2017
Arizona procedure could reduce side effects of prostate cancer treatment
PHOENIX — A new treatment for prostate cancer being used in Arizona could reduce the side effects experienced by patients, a doctor said.
Read more
Nov 28, 2017
Urologic Surgeons AZ Offer New Treatment for Prostate Cancer
Mesa Practice is First in Arizona to Use Minimally Invasive HIFU Procedure, Presents Low Risk of Impotence, Incontinence to Patients
Read more
Nov 14, 2017
Urologic Surgeons of Arizona to Offer New Option to Prostate Cancer Patients
Mesa Practice is First in Arizona to Use Minimally Invasive HIFU Procedure, Presents Low Risk of Impotence, Incontinence to Patients
Read more
Nov 14, 2017
What’s New in HIFU?
Advances in Ultrasound Technology Offer New Options to U.S. Prostate Cancer Patients
Read more
Nov 11, 2017
Deciding to Use HIFU for Prostate Cancer Treatment
According to the American Cancer Society, prostate cancer is the most common cancer among men, after skin cancer and there will be 161,360 new diagnoses this year alone.
Read more
Nov 4, 2017
Four Things Every Man Needs to Know after a Prostate Cancer Diagnosis
Men diagnosed with prostate cancer find assurance in discussing the state of their disease and treatment options in detail.
Read more
Nov 3, 2017
Dr. Bruno Nahar Interview - HIFU Prostate Treatment
Watch Dr. Bruno Nahar Interview
Nov 2, 2017
Urologic Surgeons of Arizona offers new treatment for prostate cancer
Urologic Surgeons of Arizona offers new treatment for prostate cancer
Read more
Oct 31, 2017
Calls for Early Prostate Cancer Screening of Black Men May Get a Boost From HIFU Technology
African American men who delay testing or treatment for prostate cancer run greater risks with their health than other males
Read more
Oct 9, 2017
Dr. Brian J. Miles interview
Dr. Brian J. Miles, Professor of Urology at Weill Cornell Medical College of Cornell University at Houston Methodist Hospital and specializing in urologic oncology, especially cancer of the prostate.
Read more
Sep 11, 2017
EDAP Announces Filing of New 510K
EDAP Announces Filing of New 510(k) Application for Focal One(R) Device
Read more
Sep 1, 2017
Vituro Health Adds Ablatherm Robotic HIFU
EDAP Partners with Leading US HIFU provider, Vituro Health Adds Ablatherm Robotic HIFU
Read more
May 22, 2017
New Reimbursement Code for HIFU Ablation
EDAP Announces CMS Approval of New Reimbursement Code for HIFU Ablation of the Prostate
Read more
Feb 27, 2017
Ablatherm at University of Minnesota
EDAP's Ablatherm
® Robotic HIFU Treatments Performed at University of Minnesota
Read more
Jan 23, 2017
EDAP to Exclusively Distribute LABORIE's Medical Measurement Systems
EDAP to Exclusively Distribute LABORIE's Medical Measurement Systems Urodynamic Products in Japan
Read more
Dec 19, 2016
Superior Outcomes of Focal Ablatherm-HIFU
EDAP Reports Superior Outcomes of Focal Ablatherm-HIFU Compared to Robotic Radical Prostatectomy
Read more
Dec 12, 2016
Focal One(R) HIFU Sale to Foch Hospital, Paris
EDAP Announces Focal One(R) HIFU Sale to Foch Hospital, Paris
Read more
Dec 8, 2016
EDAP Receives the Prestigious Prix Galien
EDAP Receives the Prestigious Prix Galien for its Focal One(R) HIFU device
Read more
Oct 13, 2016
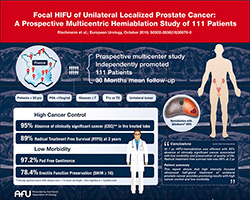
Ablatherm(R) Robotic HIFU Positioned as Efficient Option for Unilateral Prostate Ablation
EDAP's Ablatherm(R) Robotic HIFU Positioned as Efficient Option for Unilateral Prostate Ablation in Breakthrough Multicenter Study Published in Major Medical Journal
Read more
Sep 12, 2016
Ablatherm Robotic(R) HIFU at Leading Academic Medical Center
EDAP Installs Ablatherm Robotic(R) HIFU at Leading Academic Medical Center
Read more
Aug 22, 2016
First HIFU patient treated at Henry Ford Hospital in Detroit
EDAP's Ablatherm
® Robotic HIFU Treats First Patient at Henry Ford Hospital in Detroit
Read more
Jun 14, 2016
First HIFU patient treated at Allied Urological Services in New York City
First HIFU patient treated at Allied Urological Services in New York City with the Ablatherm
® Robotic HIFU device
Read more
Jan 25, 2016
First Prostate Ablation Treatments Performed Using EDAP's FDA Approved Ablatherm® Robotic HIFU
First Prostate Ablation Treatments Performed Using EDAP's FDA Approved Ablatherm® Robotic HIFU at Sylvester Comprehensive Cancer Center, in Miami, Florida
Read more
Jan 19, 2016
EDAP Announces Results of World's First Focal HIFU Trial
EDAP Announces Results of World's First Focal HIFU Trial
Read more
Dec 23, 2015
EDAP Reports Sale of Ablatherm Robotic HIFU to Sylvester Comprehensive Cancer Center
EDAP Reports Sale of Ablatherm Robotic HIFU to Sylvester Comprehensive Cancer Center
Read more
Dec 11, 2015
EDAP Receives Russian Regulatory Approval for Focal One® HIFU Device
EDAP Receives Russian Regulatory Approval for Focal One
® HIFU Device
Read more
Dec 9, 2015
Installation of Ablatherm® Robotic HIFU at University of Southern California
EDAP Reports Installation of Ablatherm
® Robotic HIFU at University of Southern California
Read more
Nov 12, 2015
EDAP Records First Three U.S. Orders for Ablatherm HIFU
EDAP Records First Three U.S. Orders for Ablatherm HIFU
Read more
Nov 9, 2015
Ablatherm FDA approved
EDAP Receives FDA Approval for Ablatherm HIFU
Read more
Sep 24, 2015
Focal One® HIFU Device Adopted by Medipole Group in France
EDAP's Focal One® HIFU Device Adopted by Medipole Group in France Read more
Jul 7, 2015
Focal One device in Switzerland
EDAP's Focal One® HIFU Device Adopted by Two University Hospitals in Switzerland
Read more
Jun 9, 2015
Focal One® HIFU at Germany's Martini-Klinik
EDAP Announces Focal One® HIFU Installation at Germany's Martini-Klinik
Read more
Mar 31, 2015
First Focal One® HIFU Device in North America First Focal One® HIFU Device in North America
EDAP Announces Sale of First Focal One® HIFU Device in North America
Read more
Mar 26, 2015
EDAP to Participate in Major
EDAP to Participate in Major, Multi-Partner Liver Cancer Development Project
Read more
Mar 9, 2015
EDAP Announces Plans to Pursue Direct De Novo 510(k)
EDAP Announces Plans to Pursue Direct De Novo 510(k) Petition in Lieu of PMA for Its Ablatherm HIFU
Read more
Feb 26, 2015
EDAP TMS SA today announced a multi-national strategic OEM partnership with Quanta System
EDAP TMS SA, today announced a multi-national strategic OEM partnership with Quanta System
Read more
Jan 9, 2015
Focal One HIFU Device Approved by Health Canada
EDAP's Focal One HIFU Device Approved by Health Canada
Read more
Nov 20, 2014
EDAP Reports Continued Strong Growth in Third Quarter 2014
EDAP Reports Continued Strong Growth in Third Quarter 2014
Read more
Nov 6, 2014
EDAP Receives FDA Guidance on PMA for Ablatherm-HIFU
EDAP Receives FDA Guidance on PMA for Ablatherm-HIFU
Read more
Oct 21, 2014
EDAP Announces Sale of Focal One Nantes
EDAP Announces Sale of Focal One(R) HIFU Device to Nantes-Atlantis Urology Clinic in France
Read more
Oct 7, 2014
EDAP Announces Sale of Focal One HIFU Device to Mannheim
EDAP Announces Sale of Focal One
® HIFU Device to Mannheim University Hospital, Germany
Read more
Sep 26, 2014
Ablatherm HIFU Technology Highlighted
EDAP's Ablatherm® HIFU Technology Highlighted at the World Congress of Endourology, Taiwan
Read more
Jun 27, 2014
EDAP Completes FDA Inspection of Manufacturing Site
FDA Inspector Reported no Findings From the Scheduled Facility Audit.
Read more
Apr 21, 2014
Reimbursement of HIFU
EDAP TMS Announces Reimbursement of HIFU Treatment for Prostate Cancer by France's Ministry of Health
Read more
Mar 20, 2014
EDAP Completes FDA Q&A Session
EDAP Completes FDA Q&A Session for Its Ablatherm-HIFU Pre-Market Approval Application
Read more
Feb 11, 2014
NEWS-First Focal One's treatments in Poland
EDAP TMS Delivers Focal One
® Device for Focal Treatment of Prostate Cancer to Hospital in Poland.
Read more
Oct 4, 2013
Arnaud Montebourg en visite à EDAP TMS
Arnaud MONTEBOURG, Ministre du redressement productif se rendra à la société EDAP TMS vendredi 4 octobre.
Read more




















.png)